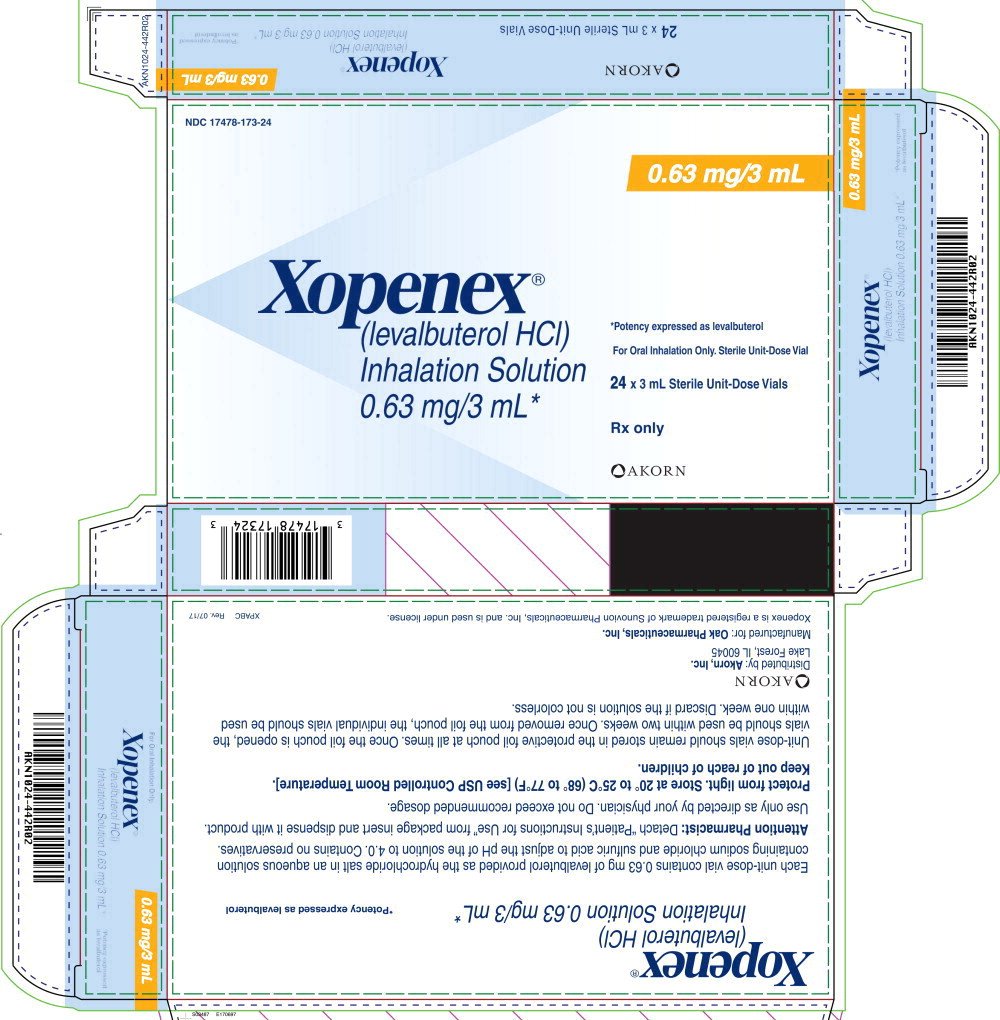
Mario Cortina - Senior Multi-Specialty Portfolio Specialist - Sumitomo Pharma America (formerly Sunovion Pharmaceuticals | LinkedIn

Sunovion Pharmaceuticals Inc. - Sunovion Announces Lonhala™ Magnair™ (glycopyrrolate) Inhalation Solution Now Available in the U.S. for the Treatment of COPD

Deceptive Promotions Used by Sunovion to Hype Respiratory Drug Brovana, FDA Charges - Parker Waichman LLP

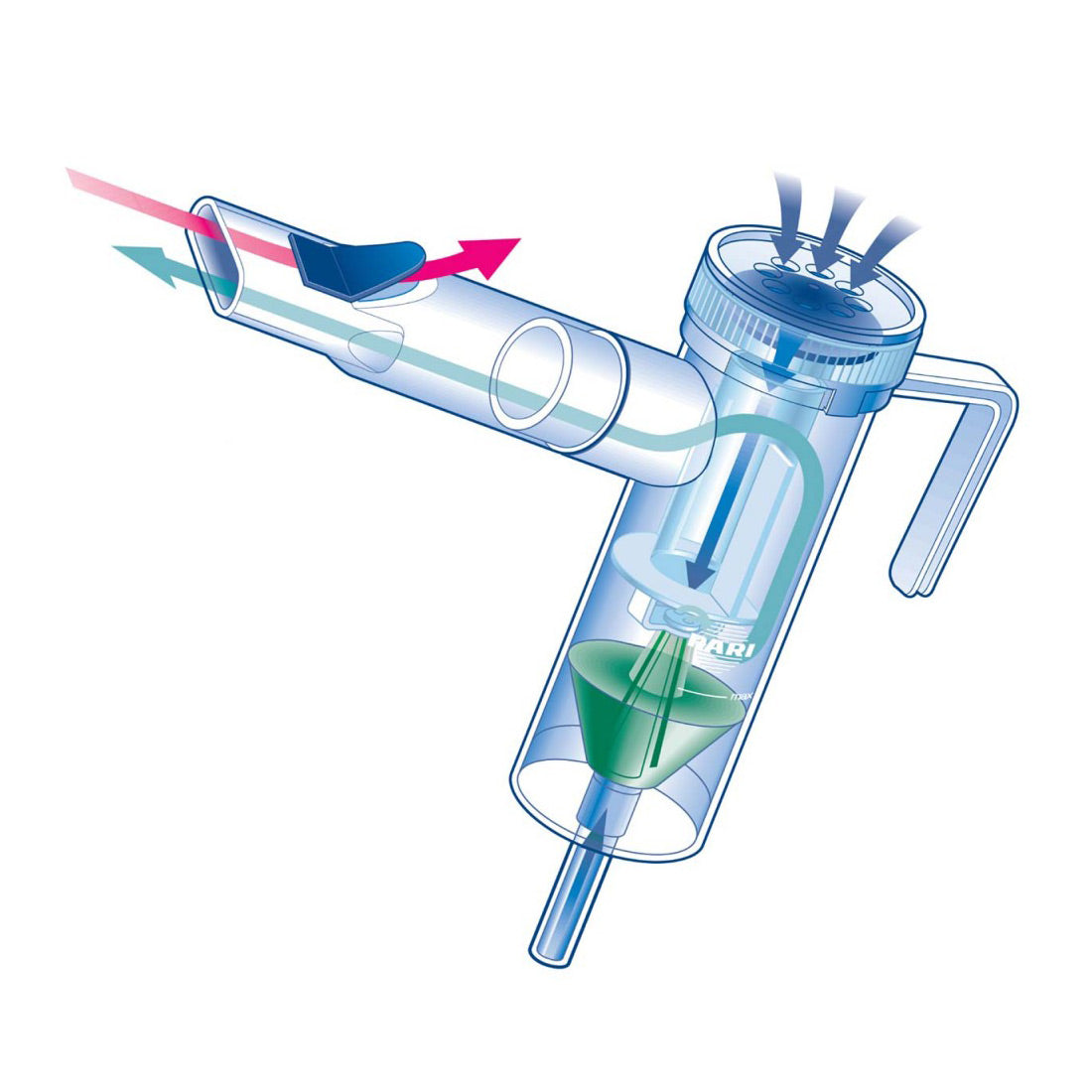
Efficacy and safety of nebulized glycopyrrolate for administration using a high efficiency nebulizer in patients with chronic obstructive pulmonary disease - Leaker - 2015 - British Journal of Clinical Pharmacology - Wiley Online Library

Sunovion Receives FDA Approval for Lonhala™ Magnair™ Inhalation Solution to Treat COPD | Business Wire

Sunovion Pharmaceuticals Inc. - Sunovion Receives FDA Approval for Lonhala™ Magnair™ Inhalation Solution to Treat COPD