Nevro receives CE mark for full-body MRI conditional labelling with the Senza system - NeuroNews International
1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the SENZA® Neuromodulation Systems
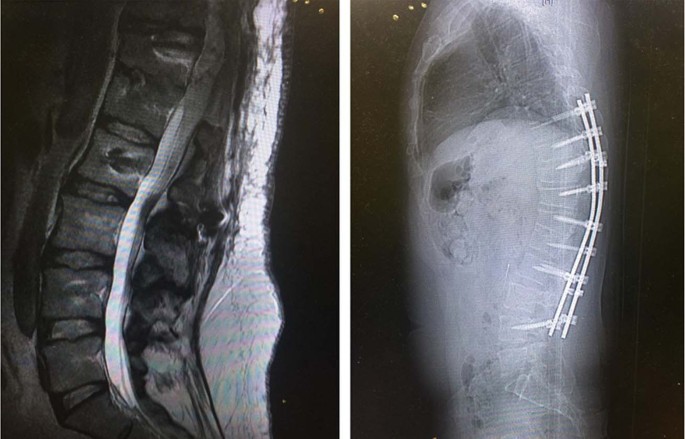
Safety and Utility of Spinal Magnetic Resonance Imaging in Patients with High-Frequency Spinal Cord Stimulators: A Prospective S
UW Health Radiology Guidelines for MRI in Patients with Common Complex Devices: Available Hours, Personnel Req
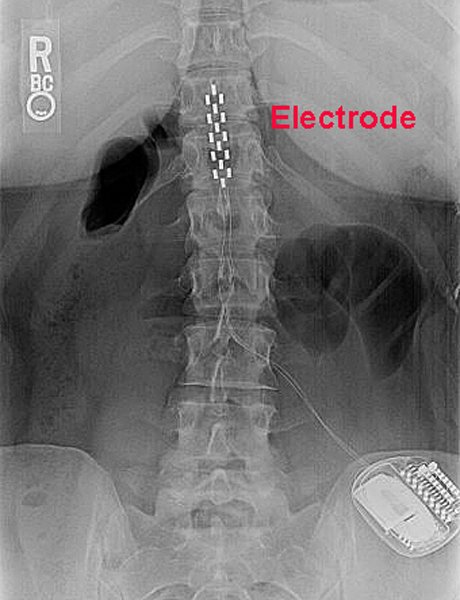
Spinal cord stimulation and rehabilitation in an individual with chronic complete L1 paraplegia due to a conus medullaris injury: motor and functional outcomes at 18 months | Spinal Cord Series and Cases

New FDA Approval Gives Medtronic Full Portfolio Of MR-Conditional Neurostim Products | Orthopedic Design Technology

A Comprehensive Practice Guideline for Magnetic Resonance Imaging Compatibility in Implanted Neuromodulation Devices - Sayed - 2020 - Neuromodulation: Technology at the Neural Interface - Wiley Online Library