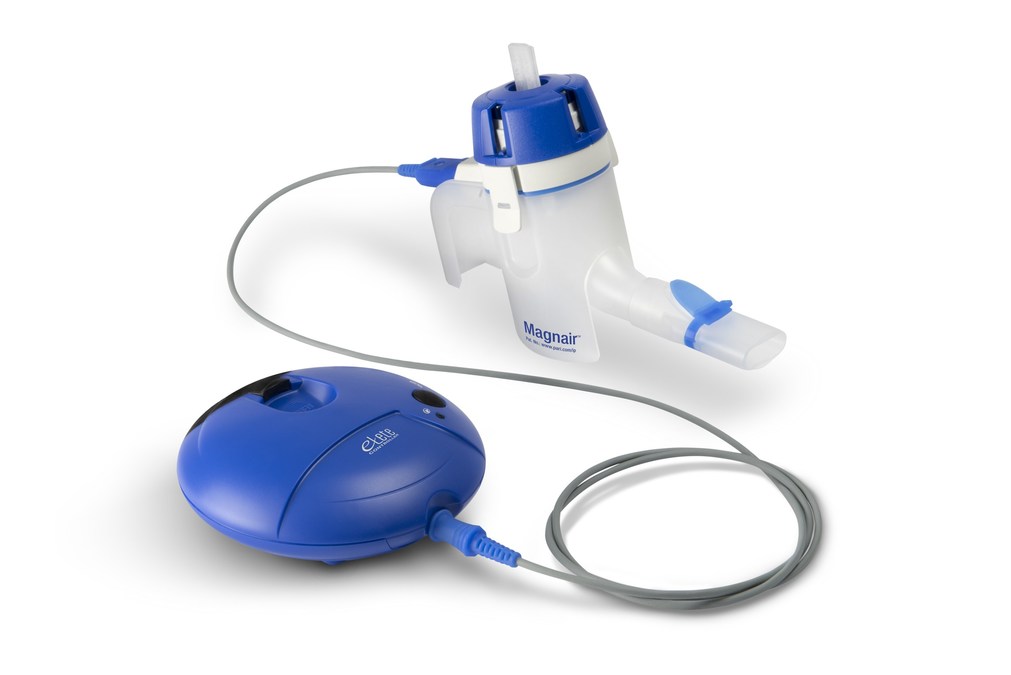
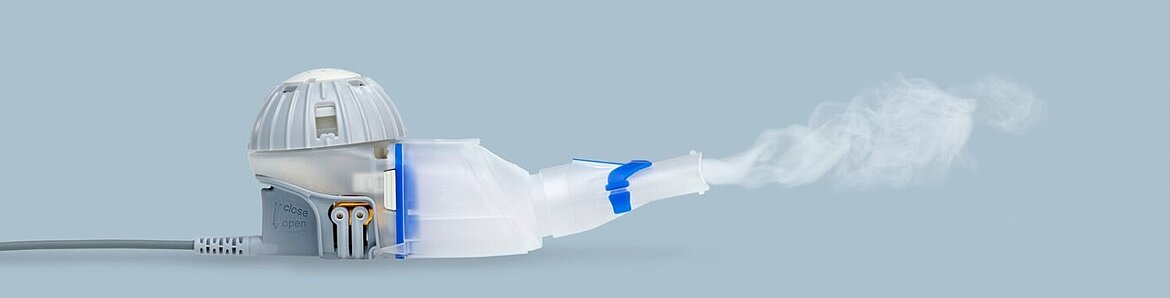
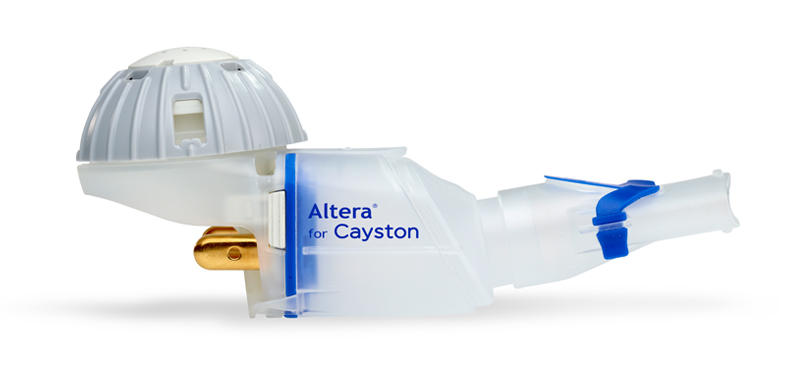
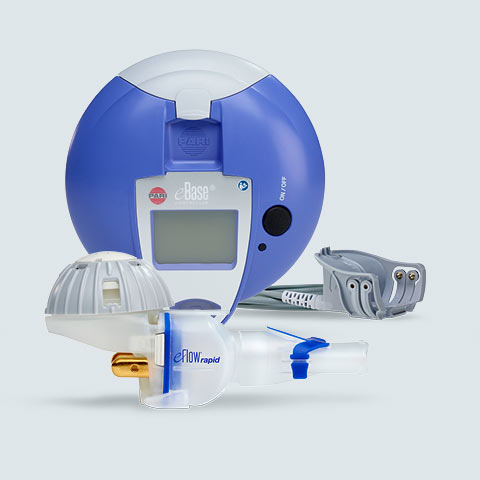
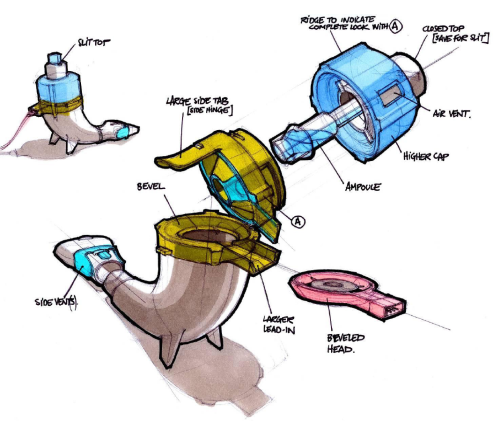
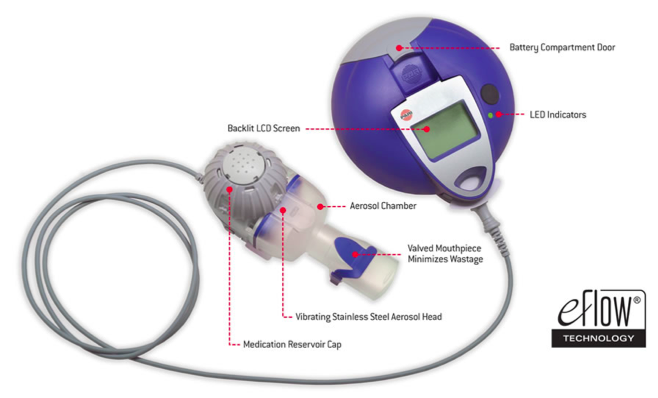
In Vitro Characterization of the eFlow Closed System Nebulizer with Glycopyrrolate Inhalation Solution | Journal of Aerosol Medicine and Pulmonary Drug Delivery
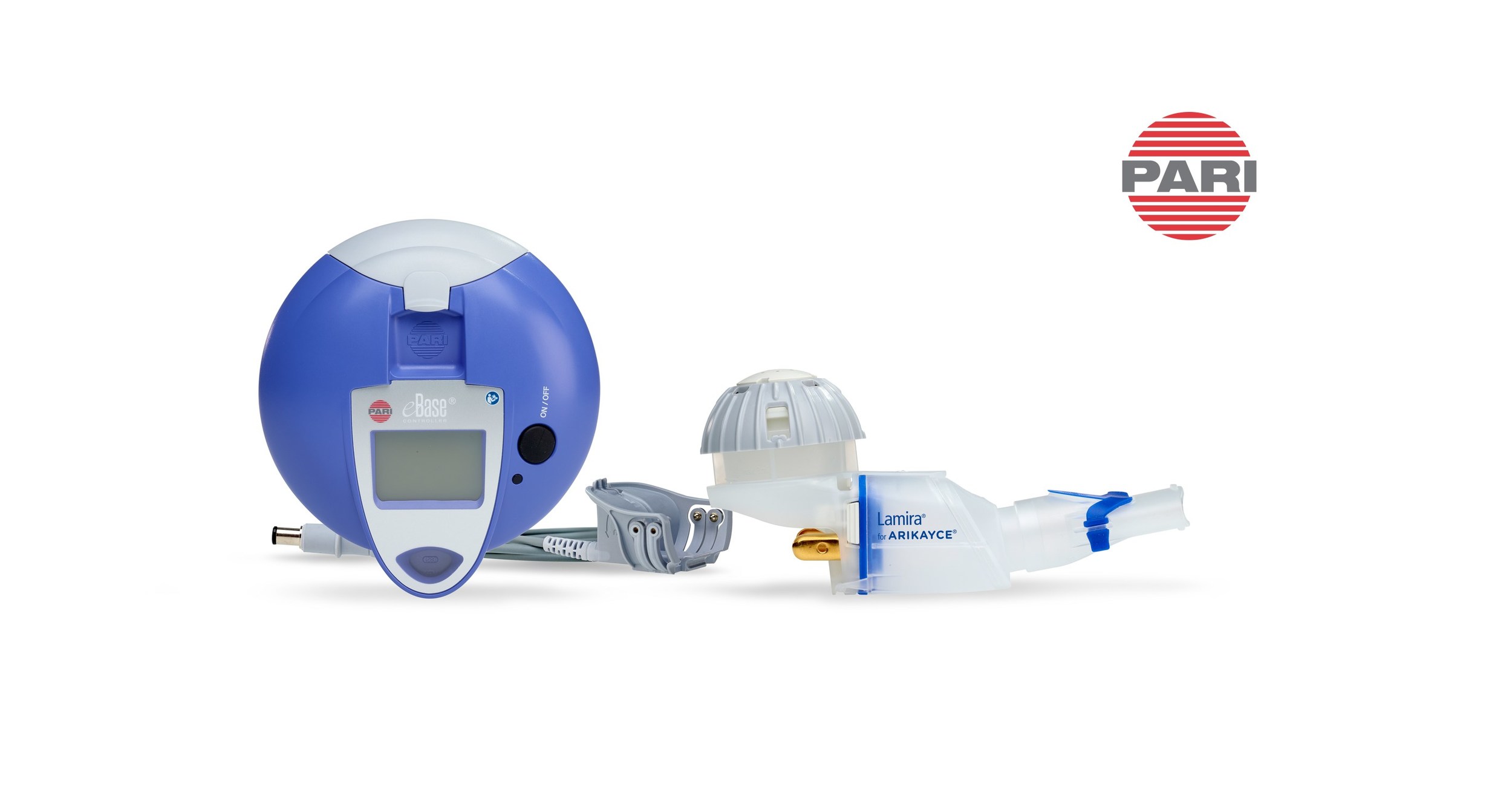
LAMIRA®, an eFlow® Technology nebulizer, expected to launch in Japan as a medical device for exclusive administration of ARIKAYCE® (amikacin liposome inhalation suspension)